| Types of
Chemical Reactions and Solution Stoichiometry |
|
| Determining Molarity: |
 |
|
Molarity and Volume:
(M1 is the molarity of solution one, which has a volume of V1.
M2 is the molarity of solution two, which has a volume of V2.) |
M1V1 = M2V2 |
|
^_Back to Top_^ |
| Gas Laws |
|
The Ideal Gas Law:
(P = pressure in atm, V = volume in liters, n = moles, R = 0.082058 L*atm/mol*K,
T = temperature in Kelvins) |
PV = nRT |
|
| Boyle's Law: |
PV = K |
|
| Charles Law: |
K = V/T |
|
Derivative of the Ideal Gas Law:
(MW = molecular weight, d = density) |
MW = dRT / P |
|
Root Mean Square:
(R = 8.3145 J/ K * mol, M = mass of a mole of the gas in kg) |
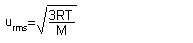 |
|
| Rate of Effusion: |
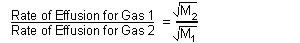 |
|
| Van der Waal's Equation: |
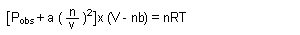 |
|
| ^_Back to Top_^ |
| Thermochemistry: |
|
| (q = heat at constant temp) |
D E = q + w |
|
| (w = work) |
W = -PDV |
|
|
DH = DE + D(PV) |
|
| c = heat capacity |
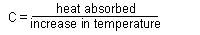 |
|
s = specific heat capacity
m = mass of solution |
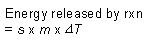 |
|
Relationship between frequency and wavelength
(c = 2.99979 x 108 m/s) |
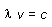 |
|
| (n = integer, h = Planck's constant = 6.626 x 10-34
J*s) |
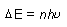 |
|
| Energy per photon: |
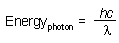 |
|
| Equation for special theory of relativity: |
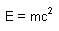 |
|
| Calculating Zeff |
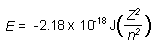 |
|
| de Broglie's equation: |
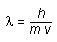 |
|
^_Back to Top_^ |
| Bonding: |
|
| (Q is charge of ions, r shortest distance between centers of
the cations and anions) |
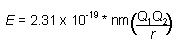 |
|
Calculating lattice energy
(k is proportionality constant, Q is charge of ions, r = shortest distance
between centers of the cations and anions) |
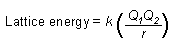 |
|
|
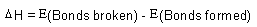 |
|
| Bond Order: |
 |
|
| Formal Charge: |
 |
|
^_Back to Top_^ |
| Liquids and
Solids: |
Clausius-Clapeyron equation
(R = 8.3145 J/K mol) |
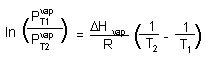 |
|
^_Back to Top_^ |
| Solutions: |
| Molality: |
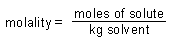 |
|
Psoln: vapor pressure of the solution
Xsolvent: mole fraction of solvent
Psolvent: vapro pressure of pure solvent |
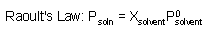 |
|
| Mass Percent: |
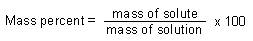 |
|
| Modified osmotic pressure formula for electrolytes |
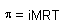 |
|
Henry's Law
P: partial pressure of gaseous solute
C: concentration of dissolved gas
k: constant |
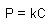 |
|
Boiling point elevation
m: molality
Kb: constant |
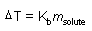 |
|
Freezing Point Depression
m: molality
Kf: constant |
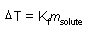 |
|
p: osmotic pressure
M: molarity
R: 0.082616 L*atm/K*mol |
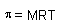 |
|
| van't Hoff factor |
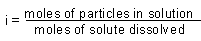 |
|
Modified for electrolyte solutions
m: molality
K: freezing/boiling point constant |
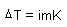 |
|
|
^_Back to Top_^ |
| Chemical
Kinetics: |
| |
Zero Order |
First Order |
Second Order |
| Rate Law |
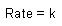 |
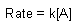 |
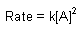 |
| Integrated Rate Law |
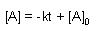 |
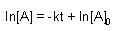 |
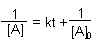 |
| Half-life |
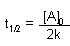 |
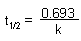 |
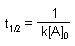 |
|
| General Rate Law: |
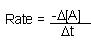 |
|
Arrhenius Equation
the whole e^(stuff) equation represents the fraction of collisions with sufficient energy
to produce a rxn |
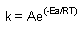 |
|
| Derived Arrhenius Equation |
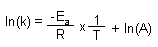 |
|
^_Back to Top_^ |
| Chemical
Equilibrium: |
|
| Calculating Equilibrium Constant: |
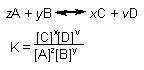 |
|
| Dn = coefficients of gaseous
products - coefficients gaseous reactants |
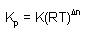 |
|
|
^_Back to Top_^ |
| Acids and
Bases: |
|
Equilibrium Expressions:
HA <--> H+ + A- |
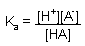 |
|
|
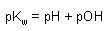 |
|
|
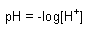 |
|
|
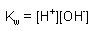 |
|
| % Dissociation |
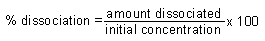 |
|
^_Back to Top_^ |
| Spontaneity,
Gibbs Free Energy: |
|
|
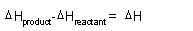 |
|
|
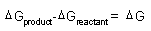 |
|
|
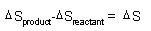 |
|
| Gibbs Free Energy: |
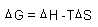 |
|
|
 |
|
|
 |
|
|
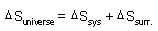 |
|
|
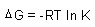 |
|
|
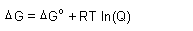 |
|
^_Back to Top_^ |
| Electrochemistry: |
|
|
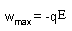 |
|
| n = number of moles of electrons transferred |
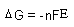 |
|
Nernst Equation
F = faraday's constant
n = number of electrons |
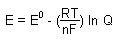 |
|