|
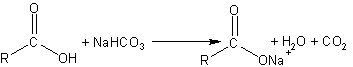 Formation of carboxylate anions |
|
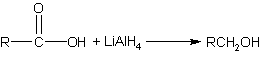 Reduction of carboxylic acids to primary alcohol
(Lithium aluminum hydride is the only effective reagent for reducing acids to
primary alcohols) |
|
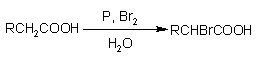 Alpha bromination of alkanoic acids
(the final product is a-bromo carboxylic acid |
|
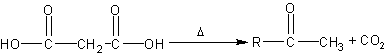 Decarboxylation of B-keto
acids to form ketones |
|
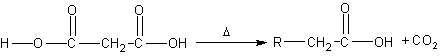 Decarboxylation makes the malonic acid lose a carbon
dioxide group |
|
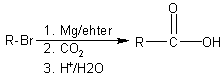 Carboxylation of organometallics
(Alkyl and aryl bromides are converted into Grignard reagents which add to the carbonyl
group of carbon dioxide. Adding acid then makes the acid group. |
|
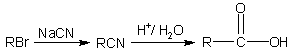 Getting carboxylic acids from nitriles |
|
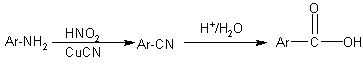 Getting carboxylic acids from nitriles |
|