Substituents and cis/transI decided to break down the
"complicated" substituents into a separate chapter. So here it goes:
Remembering what you learned from the last chapter, you should be able to name long
strands of hydrocarbon molecules. The problem arises when you encounter a molecule such as
this:
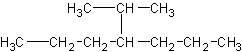
The longest parent chain is 7 carbons (heptane). But how the heck do you name the
substituent? NO, it is NOT just "butyl"
4-butylheptane would look like this:
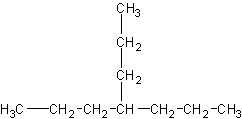
The little substituent group 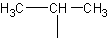 causes a lot of problems for beginners. Unfortunately, there
is no way to reason how to name this. There are a whole class of substituents which you
must memorize. Here they are: (in no particular order)
causes a lot of problems for beginners. Unfortunately, there
is no way to reason how to name this. There are a whole class of substituents which you
must memorize. Here they are: (in no particular order)
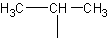
isopropyl
|
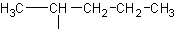
sec-butyl |
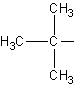
t-Butyl
(tert-butyl) |
The sec referes to the fact that the carbon that serves as the
"bridge" to the main chain is a secondary carbon (before briding to the main
chain). A secondary carbon is a carbon which has 2 carbons attached to it directly.
Primary refers to only having one carbon directly attached, and tertiary refers to three
carbons directly attached to the carbon of interest. See above for examples.
When writing two different substituents, there are two positions possible between them.
Cis and trans. Cis is "same side" and trans
is "different side." In branched alkanes, there is free rotation. The only
alkanes which get the cis/trans prefixes are the cyclic rings since they don't have free
rotation. Look at these molecules:
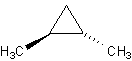
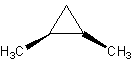
The black arrow designates that the methyl- group is sticking out (look at it 3-D). The
dashes represent going into the monitor. The one on the left is trans, the one on the
right is cis.

Free rotation: You can rotate that bromine group to become on the same side as
chlorine. That is why you never use cis/trans with chained alkanes. They can all rotate.
Although it's hard to describe, you'll have to take my word that single bonds like those
can rotate. If you want to prove it to yourself, go borrow a kit where you can build
atoms. You'll see.