Nuclear Chemistry
By Hao ZhangCharges of Elemental Components
Of the three elemental components, a proton has a positive charge, a neutron has a
neutral charge, and the electron has a negative charge.
Atomic Definitions
The atomic number (designated by the symbol Z)
represents the number of protons in the nucleus. In an element, which never has a charge,
the number of protons equals the number of electrons. The mass number (A)
is the sum of neutrons and protons in the nucleus. Therefore, A - Z
equals the number of neutrons in a nucleus.
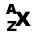
Typical Nuclide(where X equals any element):
Two Types of Atomic Stability
Kinetic stability- the likelihood of a nucleus to change into a more
stable state.
Thermodynamic stability- A comparison of the potential energy of the
nucleus to the sum of the potential energy of its protons and neutrons.
A nuclide becomes more stable through radioactive decay.
Alpha particle (a ) decay-emission of helium particle:
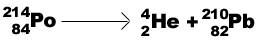
Beta particle (b -)decay- emission of an
electron:
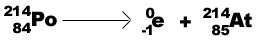
Positron (b +) emission-a positively charged
electron:
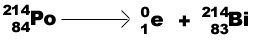
Electron capture (b -)-a gaining of electrons:
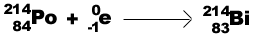
When a positron and an electron are combined, the result is 2 gamma rays. This process
is called annihilation.
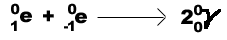
Gamma ray (g )-high energy photons that
accompany nuclear decays and particle reactions. Since they have neither mass nor charge,
they are usually disregarded in equations.

*Gamma rays are only produced in specific reactions.
Some nuclides exhibit a "zone of stability" where
radioactive decay does not take place.
- Light nuclides with proton/neutron ratio of 1 are stable.
- Even number of protons and neutrons are more stable than odd numbered.
- Special number of protons and neutrons are more stable. The "magic numbers"
are the following: 2, 8, 10, 28, 50, 82, and 126.
- All nuclides with Z = 84 are unstable.
Rates of Decay
All radioactive decay reactions are 1st order.
For a 1st order reaction:
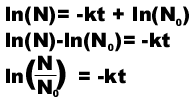
The half-life equation for a nuclear reaction
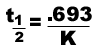
Nuclear transformation is the conversion of one
element to another.
Two methods of nuclear transformation:
1. Cyclotron - A particle is accelerated through a particle accelerator and is
penetrated into the nucleus.
2. Linear Accelerator - particles accelerated by magnetic fields bombards the nucleus.
Fission isthe splitting of a heavy nuclide into two smaller nuclides.
Excess neutrons and energy is produced in the reaction. For a chain reaction, the emitted
neutrons split nearby nuclides that produce a self-sustaining reaction.
Types of Fission reactions
Subcritical: less than one neutron splits one nuclide (This
reaction will not be self-sustaining.In order to be self-sustaining, at least one neutron
must go on to split another nuclide)
Critical: one neutron splits one nuclide
Supercritical: more than one neutrons splits one nuclide
Fusion-combining two smaller nuclides to form one heavy nuclide.
Effects of Radiation
During radioactive decay, the nuclides emit radiation.
- If the nuclide has a large half-life, the decomposition process is very slow. If a
nuclide enter into a human body, it will remain there for a long period of time.
- If the nuclide has a short half-life, the rapid emission of radiation causes a shorter
period of greater damage.
Rads (radiation absorbed dose) -measurement of radiation
1 rad=10-2J / kg tissue
Rem (roentgen equivalent for men)-measures both dosage of radiation and its
effectiveness in the environment.
1 rem=#of rads * RBE
RBE-the effectiveness of radiation to cause biological damage.