States of Matter (Liquids/Solids)
By Hao Zhang4 states of matter: Liquids, Solids, Gases, and Plasma

Intermolecular forces (weakest to strongest):
The forces involved in the three states of matter (from weakest to strongest)
- Intermolecular forces
- Covalent bonding
- Ionic bonding
- Network covalent bonding
The change in phases is caused by the change of forces among the molecules.
London dispersion forces- forces between non-polar and noble gas
molecules. The molecules involved have zero dipole moment. Generally, more electrons means
greater Dispersion forces
Polarizability-as the atomic number of the atom increases, the # of
electrons and protons also increases. This increase changes the momentary dipole action.
Based on this principle, as # of molecules or atom increases, the London dispersion
forces also increase.Examples of molecules with London dispersion forces: CH4 ;
C2H4 ; He
*The London dispersion forces in C2H4 >
CH4 because ethane has more electrons.
Dipole-dipole attractions-forces between polar molecules. The positive
pole of the molecule attracts the negative pole of another molecule. This force is only 1%
as strong as a covalent or ionic bond. The strength of the force is inversely proportional
to the distance between the two molecules.
Hydrogen Bonding-the strongest intermolecular force. Its strength is
largely due to the small size of the hydrogen atom and the large polarity in the resulting
molecule.
The atoms that form hydrogen bonding are the following: Fluorine (F), Nitrogen (N), and
Oxygen (O).
Gas State:
For tutorial on gas click here.
Liquid State:
Basic properties of liquid
- Low compressibility
- High density compared to gas
- Lack of rigidity
- Viscosity-measure of resistance to flow (stronger intermolecular forces = greater
viscosity)
- Surface tension-resistance of liquid to increase its surface area (stronger
intermolecular forces = greater surface tension)
- Capillary action-the sudden rise in level of liquid in tight space
Cohesive forces-intermolecular forces between the molecules in the liquid
Adhesive forces-intermolecular forces between the liquid and the container
Water creeps up glass because…
1.Water and glass are polar
2. Molecules in glass orient themselves to attract the water molecules. (Dipole-dipole
attractions)
3. Both water and glass have strong intermolecular forces so adhesive forces equals
cohesive forces
4. Once they reach a balance, the result is a concave meniscus
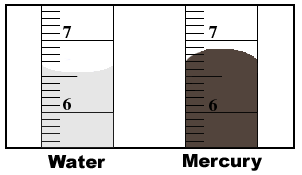
For mercury, cohesive forces> adhesive forces because there
is no attraction between mercury, nonpolar, with glass, polar. The result is a convex
meniscus.
Solids:
- Crystalline solids- organized structures of its components
- Amorphous solids- disorganized arrangement of its components
- Lattice- a 3-D representation of the components of the solid
- Unit cell- the smallest repeating unit of the lattice
Types of Crystalline solids:
| |
Atomic solids |
Molecular solid |
Ionic solid |
| Metallic |
Network |
Group 8A |
|
|
| Components that occupy the lattice points: |
Metal atoms |
Nonmetal atoms |
Group 8A atoms |
Discrete molecules |
Ions |
| Bonding: |
Delocalized covalent |
Directional covalent |
London dispersion forces |
Dipole-dipole and /or London dispersion forces |
Ionic |
Transitions between the 3 states of matter:
Fusion (melting): Solid� Liquid
Heat of fusion, D Hfus, is
enthalpy change at the melting point of the solid.
Sublimation: Solid� Gas
Evaporation: Liquid� Gas
Also known as vaporization. The energy to vaporize 1 mole of a liquid at 1 atm
is the heat of vaporization, D Hvap.
Condensation: Gas� Liquid
When a liquid is sealed in a closed container, the liquid evaporates, but at
the same time the gas condenses back to the liquid state. Equilibrium is reached when the
rate of condensation equals the rate of evaporation. The equilibrium vapor pressure is
also the vapor pressure of the liquid.
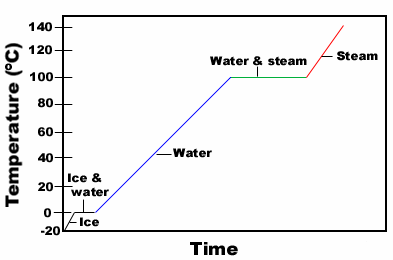
Heating Curve for water:
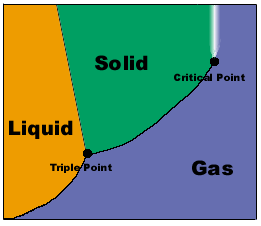
A Sample Phase Diagram
Triple point-the temperature and pressure in which all 3 states of matter
co-exist in equilibrium
Critical point-the endpoint of the liquid-gas line where no matter how much the
pressure and temperature are varied, the gas will not liquefy.
*The slope of the solid-liquid line is negative if the liquid state is denser than the
solid states.